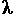
Thus hc/
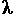 = E2-E1
= E2-E1 where h is Plancks constant 6.6E-34 J s
C is the speed of light ~3E10 m/s
If this luminescence persists then ,this phenomenon is known as phosphorescence .It is caused by the presence of metastable (long lifetime) states. To understand this fully a knowledge of some quantum mechanics is needed .Basically electrons occupy the lowest available energy level (shell) until they are excited by means of energy which raises then up to a higher energy level(because they have gained energy ).Glow in the dark materials are exhibit phosphorescence .the glow is produced by electrons falling down to the ground state and emitting their excess energy in the form of light . Because you used a red laser light source this has a lower energy then the greenish-yellow glow of the glow in the dark material (the energy is given as E=hf f is the frequency of the light and is related to the wavelength by c=f
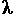 )
)The brighter glow is produced because of the laser light is so intense and contains a lot more photons then normal light ,these cause the electrons to be excited very quickly but also decay very quickly because of the their lower energy values . A laser of the type you have described is probably what’s known as a class I or maybe it is a class II laser but it will have an output power of less then 1mW ,so thermal effects are negligible.