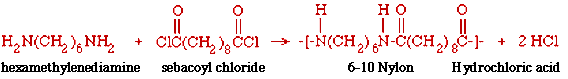
| MadSci Network: Chemistry |
A standard demonstration of the reaction that makes Nylon (a polyamide) uses hexamethylenediamine and sebacoyl chloride. The product is called 6-10 Nylon and consists of alternating units contributed by the hexamethylened iamine (6 carbon atoms) and the sebacoyl chloride (10 carbon atoms).
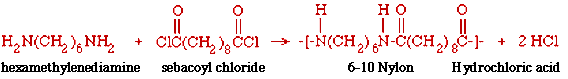
The name comes from the number of carbon atoms in the repeating units, 6 and 10 in this case. There are other types of Nylons in which the repeating units contain numbers of carbon atoms different from 6 and 10. For example, an important commercial Nylon is called '6-6' in which each repeating unit contains 6 carbon atoms. Incidently, the repeating units also contain hydrogen (H), nitrogen (N), and oxygen atoms (O) in addition to carbon (C) atoms.
The "slimy wet stuff" becomes Nylon fibers when it is dried and stretched while it is cold. This is called cold drawing of the fibers.
The science and technology of Nylon is a large subject. You can find out more about the Nylon demonstration by reading reference (1). Reference (2) has an interesting story about the discovery of Nylon. You can also learn more about Nylon in an encyclopaedia or see what your library has on the subject.
References:
1) Bassam Z. Shakashiri, Chemical Demonstrations, A Handbook for Teachers of Chemistry, vol. 1, The University of Wisconsin Press, 1983, pages 213-215, ISBN:0-299-08890-1. 2) Royston M. Roberts, Serendipity, Accidental Discoveries In Science, John Wiley & Sons, 1989, pages 170-176, ISBN:0-471-60203-5.Good luck!!