Re: What are the properties and applications of bucky balls?
Date: Mon Jun 1 08:48:01 1998
Posted By: Dan Berger, Faculty Chemistry/Science, Bluffton College
Area of science: Chemistry
ID: 895826557.Ch
Message:
 What are the properties
and applications of bucky balls?
What are the properties
and applications of bucky balls?
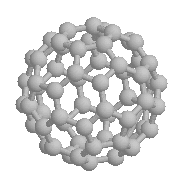
Fullerenes, an allotrope of carbon, are hollow spheres and ovoids. You
would expect them to have some properties of molecule-sized ball bearings,
and they do; for example, individual C60
molecules rota
te very rapidly in the solid state.
Fullerenes are really neat molecules, and have been/are being investigated
mostly because of that fact! Indeed, the 1996 Nobel
Prize in Chemistry was awarded primarily because fullerenes are such
nifty beasts. However, there are some potential applications of fullerenes.
- Because fullerenes are very large graphitic systems, they can
easily accommodate extra electrons. When you add three electrons to C60 you get ionic solids of the general formula
A3C60,
where A is any metal in Group I (lithium, sodium, potassium, rubidium,
cesium). These materials are actually metals,
and display sup
erconductivity at somewhat low temperatures. Current research is aimed
at getting the maximum superconducting temperature (or Tc) to higher values.
- C60 is just the right size to
fit into the activ
e cavity of HIV Protease, an enzyme important to the activity of the
virus which causes AIDS. Cramming a buckyball into the active cavity would
deactivate the enzyme and kill the virus. Ways of getting the molecule to
the enzyme are under investigation.
Most of this information has been drawn from
You should also check out the NOVA film, "Race to Catch a Buckyball."
Current Queue |
Current Queue for Chemistry |
Chemistry archives
Try the links in the MadSci Library for more information on Chemistry.
MadSci Home | Information |
Search |
Random Knowledge Generator |
MadSci Archives |
Mad Library | MAD Labs |
MAD FAQs |
Ask a ? |
Join Us! |
Help Support MadSci
MadSci Network,
webadmin@www.madsci.org
© 1995-1998. All rights reserved.
 What are the properties
and applications of bucky balls?
What are the properties
and applications of bucky balls?
 What are the properties
and applications of bucky balls?
What are the properties
and applications of bucky balls?