![]()
| MadSci Network: Chemistry |
What is the equation to determine Boyle Temperature?
pVm = RT
However, at higher pressures gases deviate from the Ideal Gas Law, following a linear expansion of the form
pVm = RT{1 + BÇp + CÇp2 + ...}
pVm = RT{1 + B/Vm + C/Vm2 + ...}
The values of the virial coefficients B, C, ... depend on temperature, and many gases have a particular temperature at which B = BÇ = 0. This temperature is called the Boyle Temperature, TB.
TB can be derived from the va n der Waals equation of state for a real gas,
![]()
In order to do that, we express the van der Waals equation as
![p = [RT/V(m)]{1/[1-b/V(m)] - a/RTV(m)}](908947075.Ch.2.gif)
(1 x)-1 = 1 + x + x2 + ...
![p = [RT/V(m)]{1 + (b - a/RT)/V(m) + ...}](908947075.Ch.3.gif)
![]()
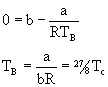
(Tc = 8a/27Rb is the critical temperature of the gas. For a derivation of the relationship of the critical constants to the van der Waals constants, see P.W. Atkins, Physical Chemistry, 3d Ed. page 31.)Thus, the Boyle temperature may be found for any gas for which the critical temperature has been determined.
| Dan Berger | |
| Bluffton College | |
| http://cs.bluffton.edu/~berger |
Try the links in the MadSci Library for more information on Chemistry.