| MadSci Network: Chemistry |
Anthony,
You have correctly identified the major component of seashells as calcium carbonate, CaCO3. Unfortunately there is not a lot of chemistry of which to take advantage. However, we can learn from the popular Easter craft of coloring eggs, whose shells are also calcium carbonate. Those shells are porous which aids in the coloring process, but the technique is the same. You can use a commerical egg-coloring kit, or simply use food coloring and follow the instructions on the box for coloring eggs. The trick is to add a little vinegar to the hot water so as to "etch" the surface of the shell so that the coloring can penetrate the crystalline structure. White shells work best.
Like all good chemists, I did the experiment myself. I had best results with red coloring and weathered shells (naturally a little porous due to the aging process). You can see the result below compared to a similar, untinted, shell on the right.
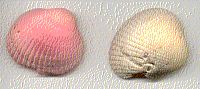
You might need to clean your shells prior to staining them. A coat of clear lacquer or varnish will protect the finished item.
You could also try a different type of ink-like dye which claims to provide a waterproof color on almost any type of surface.
Then again, you might like to purchase ready-colored shells from the many shell-craft stores on line, for example The Shell Store or Sanibel Seashell Craft Supplies.
If you've wondered about the many natural colors of seashells, for most are not white and are worth preserving in their natural state, then you might like to read "The Color of Shells".
Good luck with your crafts.
A Mad Shell-Chemist.
Try the links in the MadSci Library for more information on Chemistry.