Date: Thu Sep 4 16:28:53 2003
Posted By: Plamen Angelov, Faculty, Organic Chemistry, University of Plovdiv
Area of science: Chemistry
ID: 1061853001.Ch
Message:
Both, aldehydes and ketones, contain a carbonyl group ( that is carbon
bonded to an oxygen with a double bond: >C=O ). The reaction with hydrogen
cyanide is a typical example for addition to this group. In the course of
the reaction the hydrogen cyanide adds in such a manner that the cyano
group attaches to the carbon, and the hydrogen attaches to the oxygen of
the carbonyl group:
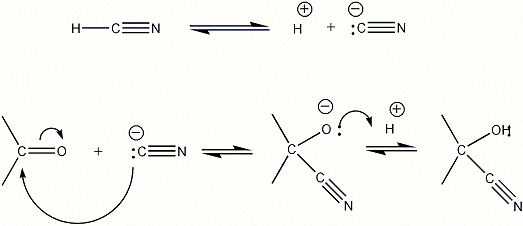 As you can see, in the end product the hydroxy and the cyano group are
both attached to the same carbon atom.
For most of the nitriles the carbon atom of the CN group is numbered as
the first atom of the main chain. Therefore, the “2” refers to the number
of the carbon atom to which the hydroxy group is attached. It is almost
always “2”, except for some complex molecules.
Here are some examples that might make the numbering clearer:
As you can see, in the end product the hydroxy and the cyano group are
both attached to the same carbon atom.
For most of the nitriles the carbon atom of the CN group is numbered as
the first atom of the main chain. Therefore, the “2” refers to the number
of the carbon atom to which the hydroxy group is attached. It is almost
always “2”, except for some complex molecules.
Here are some examples that might make the numbering clearer:
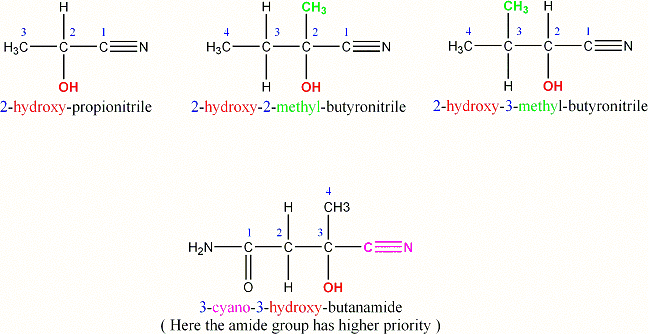 (Sometmes the 2-hydroxynitriles are also called “cyanohydrines” or alpha-
hydroxynitriles)
A very good site to acquaint yourself with the rules for naming of the
organic compounds is:
http://www.sci.ouc.bc.ca/chem/nomenclature/index-2.htm
From the above link you can learn how to determine which is the main
carbon chain in a molecule and how to number it.
(Sometmes the 2-hydroxynitriles are also called “cyanohydrines” or alpha-
hydroxynitriles)
A very good site to acquaint yourself with the rules for naming of the
organic compounds is:
http://www.sci.ouc.bc.ca/chem/nomenclature/index-2.htm
From the above link you can learn how to determine which is the main
carbon chain in a molecule and how to number it.
Current Queue |
Current Queue for Chemistry |
Chemistry archives
Try the links in the MadSci Library for more information on Chemistry.
MadSci Home | Information |
Search |
Random Knowledge Generator |
MadSci Archives |
Mad Library | MAD Labs |
MAD FAQs |
Ask a ? |
Join Us! |
Help Support MadSci
MadSci Network,
webadmin@www.madsci.org
© 1995-2003. All rights reserved.
 As you can see, in the end product the hydroxy and the cyano group are
both attached to the same carbon atom.
For most of the nitriles the carbon atom of the CN group is numbered as
the first atom of the main chain. Therefore, the “2” refers to the number
of the carbon atom to which the hydroxy group is attached. It is almost
always “2”, except for some complex molecules.
Here are some examples that might make the numbering clearer:
As you can see, in the end product the hydroxy and the cyano group are
both attached to the same carbon atom.
For most of the nitriles the carbon atom of the CN group is numbered as
the first atom of the main chain. Therefore, the “2” refers to the number
of the carbon atom to which the hydroxy group is attached. It is almost
always “2”, except for some complex molecules.
Here are some examples that might make the numbering clearer:
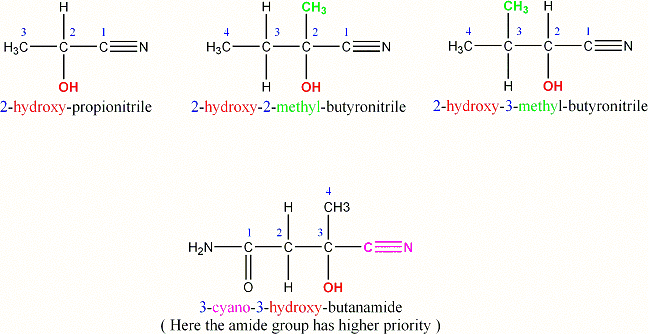 (Sometmes the 2-hydroxynitriles are also called “cyanohydrines” or alpha-
hydroxynitriles)
A very good site to acquaint yourself with the rules for naming of the
organic compounds is:
http://www.sci.ouc.bc.ca/chem/nomenclature/index-2.htm
From the above link you can learn how to determine which is the main
carbon chain in a molecule and how to number it.
(Sometmes the 2-hydroxynitriles are also called “cyanohydrines” or alpha-
hydroxynitriles)
A very good site to acquaint yourself with the rules for naming of the
organic compounds is:
http://www.sci.ouc.bc.ca/chem/nomenclature/index-2.htm
From the above link you can learn how to determine which is the main
carbon chain in a molecule and how to number it.