| MadSci Network: Biochemistry |
Hi Heather,
This is a good question. We hear about the bad effects of ultraviolet (UV) light all the time, but it isn't clear exactly what UV light does. Since you asked in particular about the effect of UV light in causing mutations (in DNA), that is all that I will discuss here, but you should also know that UV light can also have an adverse effect on proteins.
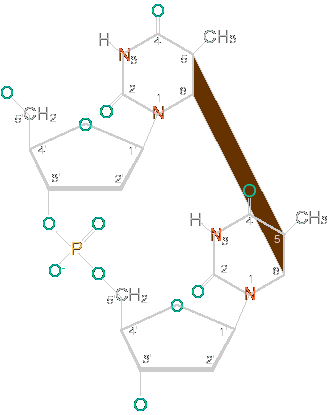
When DNA is irradiated with UV light, some of that light is absorbed by the ethylene bond in pyrmidine (thymine, T, and cytosine, C) bases in the DNA. This energy permits the formation of new bonds between adjacent pyrimidine bases, forming dimers (pairs connected by covalent bonds) of pyrimidines. Most of the time, this happens with thymines, forming thymine dimers, but cytosine dimers and thymine-cytosine heterodimers can also be formed. I've included a picture of a thymine dimer below.
| Thymine Dimer | |
 | The slanting red area represents the pair of bonds that connect the two thymine nucleotides. |
A pyrimidne dimer is a problem for several reasons. First, there is no base-pairing with the complementary purines (A or G) on the other strand of DNA. This changes the shape of the DNA molecule in the area of the dimer, which makes it difficult for the polymerase enzyme that copies DNA to move through the region of the dimer. Perhaps most importantly, beause the dimer is not making base pairs, the polymerase does not know what nucleotide to add to the new DNA strand when it reaches the dimer. It basically cannot see anything there. In some cases, the polymerase will just skip over the dimer, effectively deleting two bases from the DNA strand. In other cases, the polymerase will make a guess as to what base belongs in that position, and will incorporate something at random.
However, the cell has some systems that protect the DNA from mutations by repairing damage to the DNA (such as pyrimidine dimers). Usually, these systems (for example the UvrABCD system) will find the damage to the DNA, and will fix the damage, either by using light to break the bonds between the pyrimidines, or by cutting out the section of the DNA strand that contains the dimer and using the sequence of the opposite strand to fill in the gap. I should also point out that Yeast cells have a special polymerase (called Polymerase eta) that can take over for the regular polymerase (Polymerase delta) when Polymerase delta encounters a pyrimidine dimer, and can incorporate the correct nucleotides opposite the dimer.
When the DNA contains a large number of pyrimidine dimers, these damage repair systems go into "overdrive" in what is called the SOS response. This is the cell's "last ditch" emergency effort to repair its DNA, and it will sometimes do drastic things to save the DNA. For example, if there are too many pyrimidine dimers, the DNA repair systems may make too many cuts on both strands of DNA, and as a result, different pieces of DNA will be stuck together by the SOS response our of order.
So, UV light causes pyrimidine dimers. If there are only a few such dimers in an organism's DNA, they can easily be repaired. However, as the numbers increase and it becomes harder to repair them in a timely fashion, DNA polymerases may incorporate mistakes (mutations) into the DNA during replication. Finally, when there are an overwhelming number of dimers, the cell will enter the SOS response, and (if it survives) this can result in dramatic changes to the DNA sequence.
Here are some references that go into these issues in more detail. The papers cited below might be too involved for you, but you can find quite a lot of information about DNA replication and mutation repair in a college-level biochemistry text, such as Biochemistry by L. Stryer. Keep asking those questions!
A Gentil, F Le Page, A Margot, C W Lawrence, A Borden, and A Sarasin. (1996) Mutagenicity of a unique thymine-thymine dimer or thymine-thymine pyrimidine pyrimidone (6-4) photoproduct in mammalian cells. Nucleic Acids Res. 24: 1837–1840
Washington, M. T., Johnson, R. E., Prakash, S. and Prakash, L. (2000) Accuracy of Thymine- Thymine Dimer Bypass by Saccharomyces cerevisiae DNA Polymerase Eta. Proc. Natl. Acad. Sci U.S.A. 97: 3094-3099.
Washington, M. T., Helquist, S.A., Kool, E. T., Prakash, L. and Prakash, S. (2003) Requirement of Watson-Crick Hydrogen Bonding for DNA Synthesis by Yeast DNA Polymerase Eta. Mol. Cel. Biol. 23: 5107-5112.
Washington, M. T., Wolfle, W. T., Spratt, T. E., Prakash, L. and Prakash, S. (2003) Yeast DNA Polymerase Eta Makes Functional Contacts with the DNA Minor Groove Only at the Incoming Nucleoside Triphosphate. Proc. Natl. Acad. Sci U.S.A. 100: 5113-5118.
Try the links in the MadSci Library for more information on Biochemistry.