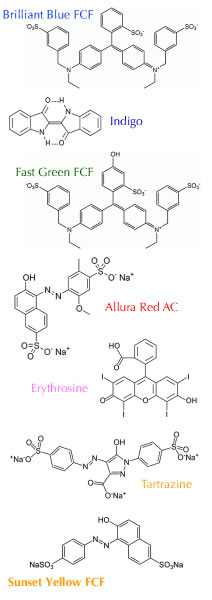
You raise a particularly interesting question, and the answer (I think) has to do with the structure of the food coloring dyes used.
Currently (according to wikipedia) there are seven dyes that are permitted in food: these are
FD&C Blue No. 1 / Brilliant Blue FCF, E133 (Blue shade)
FD&C Blue No. 2 - Indigotine, E132 (Dark Blue shade)
FD&C Green No. 3 - Fast Green FCF, E143 (Bluish green
shade)
FD&C Red No. 40 - Allura Red AC, E129 (Red shade)
FD&C Red No. 3 - Erythrosine, E127 (Pink shade)
FD&C Yellow No. 5 - Tartrazine, E102 (Yellow shade)
FD&C Yellow No. 6 - Sunset Yellow FCF, E110 (Orange
shade).
Now if you look at their structures (to the right) you will see that they all have aromatic (alternating single-double bond) rings and chains, which would normally be considered hydrophobic (water insoluble). But, because they also contain lots water soluble groups (like OH, SO) they are (somewhat) soluble in water.