Your chiropractor needs to take some basic organic and biochemistry again. Benzoate is chemically related to benzene, yes. But so (for example) are vanillin, benzaldehyde (one of the odor/flavor compounds in almonds), and aspirin. Not to mention one of the twenty essential amino acids, phenylalanine.
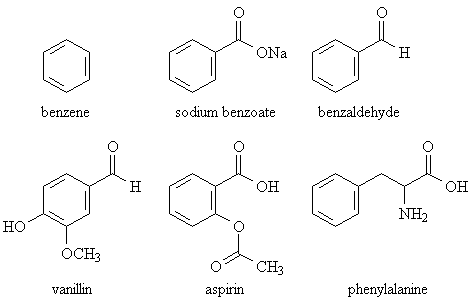
The toxicity of benzene is related to the way it is broken down in the body. None of the other compounds I've listed, including sodium benzoate, are broken down in that way. And that's just that!
For comparison, toxic doses of oral sodium benzoate tends to be in the 10-50 grams per kilogram of body-weight range, or between half-a-cup and a cup of the stuff for a 110-pound adult. For benzene, we're talking about 50 milligrams/kilogram, or about half-a-teaspoon for that same 110-pound adult.
"The dose makes the poison," and sodium benzoate is not permitted as a preservative at levels above 1 part per thousand. If your mouthwash contains large amounts of sodium benzoate, I'd be careful with it. But there's no particular need to be paranoid. It's not as if it were, say, benzene.
More information can be found in hazard.com's MSDS database.
| Dan Berger |
| Bluffton College |