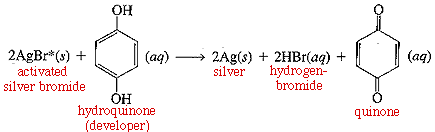
| MadSci Network: Chemistry |
Good question.. It shows that you're thinking!
Black and white photographic films contain small grains of silver salts, usually silver bromide - AgBr. When the film is exposed to light the AgBr becomes activated, like so:
AgBr + light ---> AgBr*
The development process (which I'll go into since it's an interesting
oxidation/reduction rection) leads to the formation of elemental silver
(Ag) on the plastic backing of the film, creating the dark regions
in the negative. The amount of silver depositied is direcly proportional
to the amount of light hitting a given region of film. The last
step of b&w development - fixation - removes the remaining unactivated
AgBr to give you the clear regions in the photographic negative.
However, you don't need to expose film to light to develop it - as you suggest, placing a strip of film near a radioactive source will expose the AgBr. This ability of ionizing radiation to expose common b&w film is commonly used to monitor people's exposure to radioactive sources such as 125I, or 32P. It is also the basis of how X-rays are taken of the bones.
Ionizing radiation comes in three flavors:
When a radioactive element breaks down, it emits particles and/or radiation. The result of this event is the release and transmission of energy through space in the form of a wave (radiation!). Like light waves, radioactive waves can active the AgBr in photographic film, thus exposing it. Furthermore, since some types of radioation can penetrate 'solid' materials for short distances, you don't need to directly expose the film to the radioactive source.
However, you don't 'see' anything until the film has been developed. The development process involves a series of oxidation/reduction reactions. Oxidation/reduction just means some molecules lose electrons (bceome oxidized) while others gain electrons (become reduced). The net reaction taking place is:
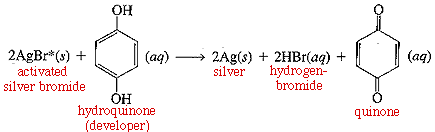
Photographic developer contains hydroquinones which will donate electrons to the activated AgBr. This reaction oxidizes the hydroquinone to quinolone, and reduces the silver ion in AgBr to elemental silver (Ag+ --> Ag). In the meantime, bromide ion is released (Br-) and can combine with loose protons (H+, or a hydrogen atom stripped of its electron, which went to silver) to form HBr.
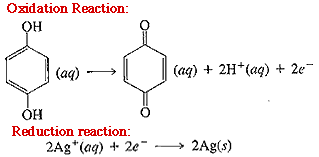
Added together, the two equations are:
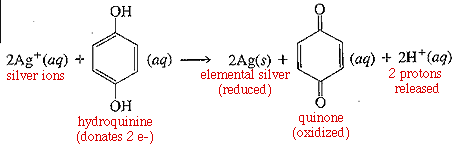
Thanks for the question.
-Nick