| MadSci Network: Environment & Ecology |
Hi Keith:
I hear people talking about lakes "inverting" a lot when I'm fishing, and it's not strictly true, a better term is "turnover".
I'll explain: The density of water (how heavy it is in a given volume)
depends on its temperature, and water is at its densest ('heaviest') at
around 4 degrees Celcius. Water that is colder or warmer than 4 degrees
will be 'lighter', and will tend to float on top (this is ultimately
caused by our good friend gravity). This property of water makes it tend
to form into layers – in a lake, the interface between those layers will
know as a thermocline (more on that in our archives here).
Because of this density effect, the densest ('heaviest',
4oC) water will always tend to lie underneath warmer, or colder
water. This is also why, if one swims downward in most lakes (especially
Canadian ones!), one will eventually encounter cold (4oC) water.
This is actually a pretty stable situation too, if water at the surface
is cooled (if it’s warmer than 4 oC) or warmed (if it’s colder
than 4oC), it will sink downwards, but once it reaches 4
oC, it cannot become heavier, and will stop sinking once it
reaches the deep water. If deep water is warmed (or cooled), it will
become lighter and tend to float upwards, but it also will stop when it
reaches water at the same temperature. Changes in temperature of deep
water can occur (say from geothermal inputs), but they are quite rare.
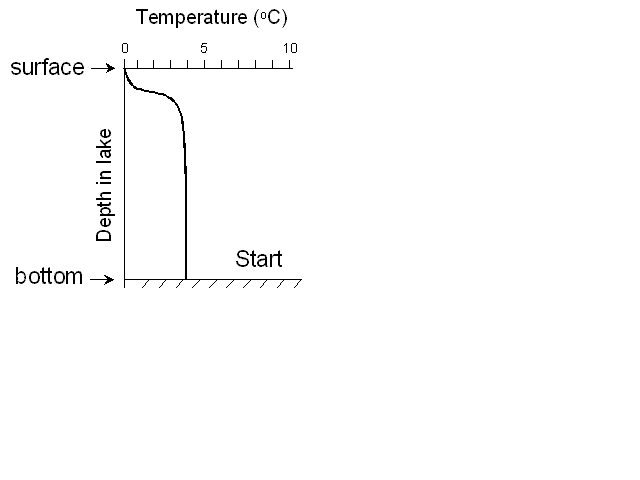
As a quick aside, I’ll explain what turnover is - think about a lake in
the winter that is covered in ice - The water directly underneath the ice
will be very close to 0oC, the temperature of the ice. Moving
down towards the bottom of the lake, the water will become warmer until it
reaches 4oC. If we plot the temperature as a function of depth
it will look like this:

In the spring, after the ice melts, the sun will begin to warm up the
water at the surface, and winds will mix it up a bit, until we reach the
situation where the surface layer is more-or-less the same temperature as
the deep layer. A made a little animation to demonstrate this:
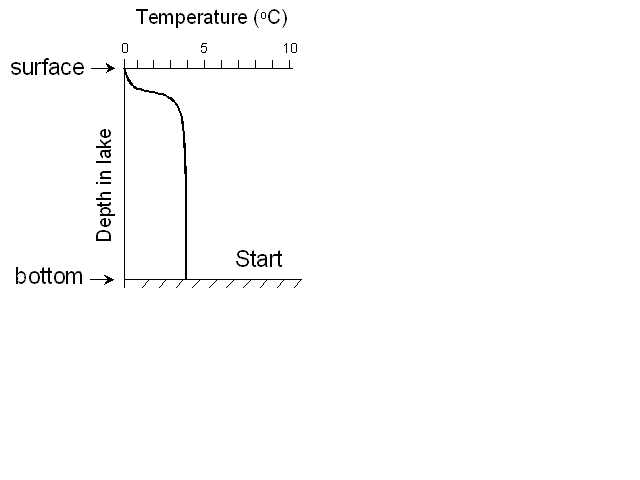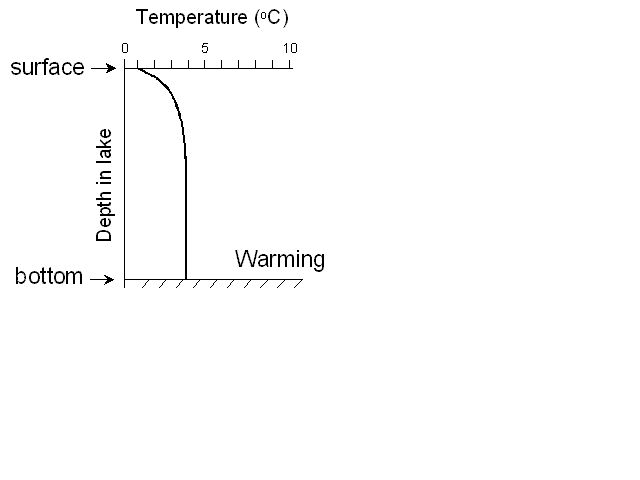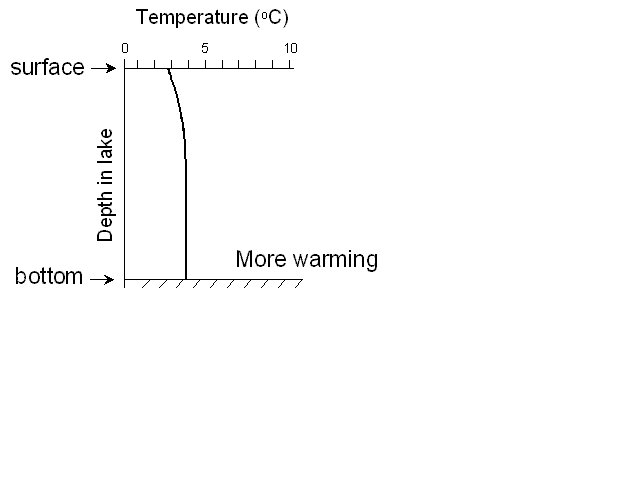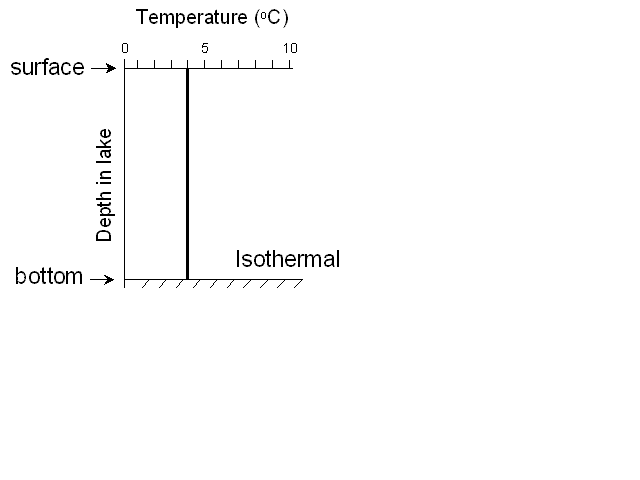
At this point we call the lake isothermal (literally “same
temperature”) – there is no difference between the surface and bottom
waters, so if the wind blows on the lake, it can mix it from top to
bottom. This is what we call turnover – when the lake can be completely mixed.
As the top layer begins to warm from continued warming at the surface,
a thermocline will eventually form (from the interplay of solar heating
and wind mixing, actually), which will tend to keep the surface waters
separated from deep waters – wind mixing will then be mostly confined to
the surface layer. In autumn, the opposite happens, as the days get
shorter and the weather cooler, the lake will lose heat from the surface
until it is again isothermal, and a fall turnover can occur.
As far as your fish kill goes, I wouldn’t suspect an inversion event, it would take some fairly serious volcanic activity underneath the lake to cause that. I would suspect that it might have been oxygen depletion. I notice that Moyie lake has a pretty big provincial campground, as well as a fair amount of development nearby, and that will mean lots of nutrient inputs. That cultural eutrophication can lead to large amounts of plankton growth. When those plankton die and sink down into the bottom water, they are broken down by bacteria, who also use oxygen. If there is enough production, it is possible for the bottom waters to run out of oxygen. For some fish with fairly narrow temperature ranges (trout are particularly sensitive to this), the surface waters can be too warm, which leaves them nowhere to go and can lead to die-offs.
I hope that helps!
Rob Campbell, MAD Scientist
Try the links in the MadSci Library for more information on Environment & Ecology.